References
Contemporary strategies and materials to protect the dental pulp
From Volume 44, Issue 8, September 2017 | Pages 731-741
Article

When returning damaged teeth to form and function, the aim of restorative dentistry must also be to preserve the heath and integrity of the dental pulp. The disease process and subsequent operative intervention will cause an inflammatory response within the pulpal tissues. It is important that the restorative dentist considers the measures that he or she can take to protect this inflamed tissue and create an environment which is conducive to healing. Traditionally, linings and bases were placed on the pulpal surfaces of cavities in an attempt to achieve this aim. However, knowledge and understanding have advanced and, with the development and availability of new dental materials, new strategies are employed in contemporary practice. This paper reviews the approach employed for many years after cavity preparation and explores how this new thinking, combined with newer dental materials, has shaped the current management of pulpal protection.
Traditional liners and bases
For many years, once cavity preparation had been completed, an intermediate material1 was placed on those surfaces overlaying the pulp prior to the placement of the definitive facilitating material. The selection of this restorative material was influenced by the proximity of the pulp following caries removal.1 These materials were crudely classified into liners or bases. Baum et al's Textbook of Operative Dentistry,1 whose ninth edition was published in 1985, defined liners as ‘materials that are placed as thin coatings, and their main function is to provide a barrier against chemical irritation. They do not function as thermal insulators. They are not used to produce a structural form for the preparation. Examples of liner materials are varnish-type materials to which calcium hydroxide or zinc oxide powder is added.’1 Bases, on the other hand, ‘function as barriers against chemical irritation, provide thermal insulation and resist forces applied during condensation of the restorative material. They are capable of being shaped and contoured to specific preparation forms. Examples of these materials are zinc oxide-eugenol, zinc phosphate, zinc polycarboxylate (polyalkenoate/polyacrylate), and glass ionomer cements and some of the commercial preparations containing calcium hydroxide.’1 Furthermore, bases were effectively a dentine replacement material used to decrease the bulk of restorative material.2 Depending on the depth of the cavity, these materials were used singly or in combination with another.
Traditional cements used as liners and bases
The dental cements which were used for this purpose had supposedly sedative properties and possibly some biocidal effects. A commonly used material was zinc eugenolate, ie set zinc oxide eugenol cement. This material had bactericidal properties, inhibiting bacterial metabolism, but could not be placed directly onto an exposed pulpal tissue as it was cytotoxic and would cause an acute inflammatory reaction.
The acid component of both zinc polycarboxylate cement and glass ionomer cement is polyacrylic acid. This is a weak acid with a relatively high molecular weight. Its long chains are not readily able to diffuse along the dentinal tubules and so do not inflict any chemical trauma on the pulp.2 The pH of all of these set cements is neutral with a low thermal diffusivity. They were used in thicknesses of at least 2 mm so that their mechanical properties were sufficient to withstand forces exerted during function. Their consistency sometimes made handling and placement difficult.2
Setting calcium hydroxide cement, such as Dycal (Dentsply, York, PA, USA) or Life (Kerr, Orange, CA, USA), was extensively used as a lining material. These products had to be placed in a thin layer as it was weak in compression. To achieve this, it was recommended that the material was air thinned using a gentle stream of air on placement2 (Figure 1). The rationale behind the use of this material was that its pH is 12.5 and this high alkalinity causes liquefaction necrosis of superficial pulp, removing up to 1.5 mm of the inflamed tissue. Neutralization of toxicity then occurs in the deep layers with coagulative necrosis, which irritates the adjacent pulp causing minor inflammation. This leads to the stimulation of the pleuripotential pulpal cells which differentiate into odontoblasts laying down a calcified dentine bridge.3 This mode of action is at odds with the basic tenet of a liner material in that its main function is to provide a barrier against chemical irritation.1 It is obvious to question the rationale of placing an irritant material to have an effect on an already inflamed tissue and whether this irritation contributed to pulpal death. Furthermore, the high solubility of these products was also of concern and cases were reported of these materials ‘disappearing’.4

Role of micro-organisms
In contemporary practice, the understanding of the importance of the role of micro-organisms is well recognized, with today's modus operandi being to decrease or eliminate microleakage. Microleakage may be defined as ‘the percolation of minute amounts of fluid, debris and micro-organisms through the microscopic space between a dental restoration or its cement and the adjacent surface of the cavity preparation’.2 It has been reported that there are between 7,000–59,000 dentinal tubules per square millimetre (mm2)5 and these may be opened when the dentine is prepared. The density of tubules increases in the vicinity of the pulp rising to between 59,000 and 76,000 per mm2.6 Furthermore, the diameter of dentine tubules is approximately 0.9 µm at the amelo-dentinal junction and 2.5 µm near the pulp.5 This creates huge potential for microbial ingress which in turn leads to pulpal inflammation.7 The ‘sensitivity’ experienced by patients was therefore not due to thermal diffusivity of the restorative material but to rapid fluid flow through the dentinal tubules. This so called hydrodynamic theory of dentine hypersensitivity was first postulated by Brännström in 1972.8 Contemporary strategies aim to seal these dentinal tubules so preventing dentinal fluid movement and microbial ingress.6 This may be achieved with a thin layer of material, by definition, a dentine-bonding agent. In a deeper cavity, where the remaining dentine thickness is less than 0.5 mm, there is more potential for micro or frank pulpal exposures to be present and so the dentist may wish to select a material which also creates a biocompatible environment to facilitate pulpal healing.3Table 1 lists the ideal properties of a lining material.
| ▪ Adheres to dentine |
| ▪ Seals dentinal tubules |
| ▪ Good physical properties (in particular compressive strength) |
| ▪ Radio-opaque |
| ▪ Command cured |
| ▪ Insoluble |
| ▪ Surface not damaged by cavity treatment such as acid etching |
Materials used in contemporary practice
Resin-modified glass ionomer cement
The material which bridges both eras is resin-modified glass ionomer cement (RMGIC), having arrived to market in the early 1990s. This is placed either onto freshly cut dentine or to cover set calcium hydroxide cement completely, in the case of a direct or indirect pulp cap, as the material is contra-indicated to be placed on exposed pulpal tissue owing to the cytotoxic effects of hydroxyethyl methacrylate (HEMA), one of its constituents2 (Figure 2). Resin-modified glass ionomer cement has many advantages. It can seal dentinal tubules, so minimizing or preventing microleakage. It adheres to tooth tissue by calcium chelation and subsidiary dentine-bonding. As its principal method of curing is by photo-initiation, it has a command cure which equates to a long working time and early strength when set. During this polymerization reaction, when its initiator decomposes, the chemical breakdown products benzoyl iodides and benzoyl bromides are released. These chemicals are cytotoxic and effective against any residual cavity bacteria.9,10 Many clinicians favour and use this material as a cavity-lining material in contemporary practice. Commercial examples of these cements include Fuji II LC (GC, Leuven, Belgium) and Vitrebond Plus (3M ESPE, St Paul, MN, USA).

Dentine-bonding agents
With the ever increasing use of restorative materials which require to be bonded by a resin such as a dentine-bonding agent, many operators have questioned the need to place any lining material at all. Such an intervening material would compromise the bond achieved between the dentine and either the resin-based composite or, by definition, bonded (sealed) amalgam.11 Dentine-bonding agents may also seal the dentinal tubules, as illustrated by the so-called intermediate dentine sealing technique,12,13,14 where a dentine-bonding agent is applied to the freshly prepared dentine of a vital tooth. The dentine-bonding agent is then cured prior to temporization or impression-taking to protect the freshly cut dentine, by preventing bacterial ingress, decreasing post-operative sensitivity and increasing the bond strength, as the collagen fibres do not collapse in the period while the restoration is being fabricated.14,15 Dentine-bonding agents commonly contain HEMA and so cannot be placed directly onto exposed pulpal tissue. For this reason, this technique may not be applicable where little dentine overlaying the pulp remains, as discussed earlier.
Flowable resin-based composites
Flowable composites are resin-based composite materials where the filler loading has been reduced or viscosity controllers have been added. These result in a lower viscosity when compared to conventional resin-based composite materials2 (Figure 3). One of the many indications for these materials is to use them as a lining material to cover the pulpal floor.16 They have the advantage of bonding to the dentine (with the help of a dentine-bonding agent) and therefore some sealing of the dentinal tubules will occur. They adapt well to the cavity walls due to their flow properties. Furthermore, if resin-based composite is being used to restore the cavity, then there will be chemical compatibility and adhesion of the flowable material to the overlaying resin-based composite. Another advantage is when a flowable resin-based composite was used as such an intermediate layer; a reduction in measured cuspal defection was observed.17 Cuspal deflection can occur during curing of the resin-based material as a result of polymerization shrinkage and can lead to cracked cusp syndrome or fracture of the tooth tissue. This is especially so in teeth with poor compliance due to stresses generated within the dental tissues.18 Furthermore, cuspal deflection can also lead to bacterial microleakage and ultimately to marginal discoloration, secondary caries and pulpal inflammation or necrosis,19 potentially manifesting in pain.

Bulk fills
The bulk fill restorative materials are an extension of flowable resin-based composite materials, also requiring to be used in combination with a dentine-bonding agent. This family of materials was developed in an attempt to try to reduce the stresses which arise in the system as a result of polymerization shrinkage. This has been achieved by altering the chemistry of the material in a number of ways, depending on the specific product.20 The first commercial product available was SDR® (Dentsply) and its low viscosity permitted the material to flow into the cavity, with a good adaptation to the floor and walls with potentially fewer voids. One bulk fill material SonicFill™ (Kerr Mfg Co) is available which is used in conjunction with a handpiece (Figure 4). This hardware applies sound waves which reduce the viscosity of the material by 87% so allowing it to be actively injected into the cavity. As its flow properties are increased so its adaptation to the cavity walls is enhanced, thereby reducing voids, which was a particular shortcoming of incremental layering.21

The indications of bulk fills are broadly similar to those of flowable resin-based composite materials16 and so can be used effectively as a lining material without the attendant problems of conventional resin composite materials.
Therapeutic restorative cements: tricalcium silicate
The last group of materials used in contemporary practice to protect the pulp after cavity preparation is the tricalcium silicate cements. These materials are based on calcium silicate chemistry and related to Mineral Trioxide Aggregate (MTA). This latter material has been used for many years in surgical endodontics as a root end filling material22 and to repair perforations,23 as well as creating a barrier for teeth with open apices.24 More recently, its use as a pulp-capping material has been investigated. ProRoot MTA (Maillfer, Dentsply), MTA-Angelus (Angelus Solucoes Odontogicas), Tri-calcium phosphate (Activa BioActive, Pulpdent) and Bio-Aggregate (Innovative Bioceramix)3 are examples of currently available commercial products. The original MTA materials had an undesirably long setting time (circa 14 hours) which precluded their use in cavities prior to their restoration. However, in 2011, Septodont (Saint-Maur-des-Fossés, France) launched a product called Biodentine™ (Figure 5) which was specifically developed to be used to protect the pulp. This cement has a working time of six minutes and is set in 12 minutes from the start of mixing.
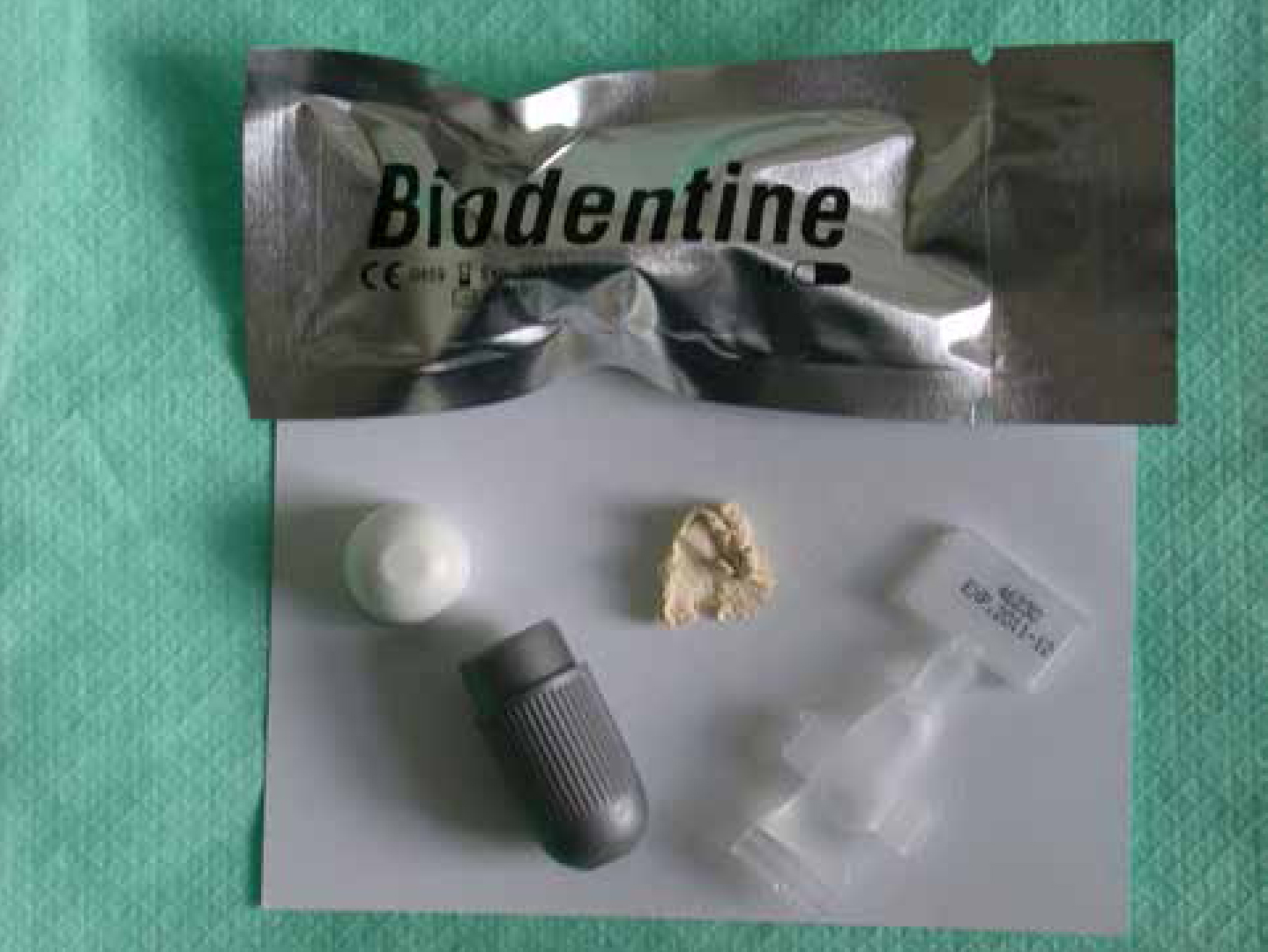
Tricalcium silicate cements are supplied as a powder and liquid which are mixed together. The powder primarily contains tricalcium silicate as the main core material and dicalcium silicate as a second core material. Calcium carbonate and calcium oxide are used as fillers, iron oxide is included to influence the shade and zirconium dioxide used as a radiopacifier. The liquid component contains a hydrosoluble polymer water-reducing agent, which acts as a superplasticizing agent to help retain the workability of the cement almost as a surfactant effect.25,26 Calcium chloride is included as an accelerator. The setting reaction is a hydration reaction and these materials may be termed hydraulic cements. During setting, the material has hydrating tricalcium silicate cement grains surrounded by a matrix of calcium silicate hydrate and calcium hydroxide, along with the presence of calcium carbonate. This latter chemical acts as a nucleating site to calcium silicate hydrates27 and may be responsible for a high pH27 and high calcium ion release.25 The high pH promotes inflammatory cell recruitment28,29and the polycarboxylate in the liquid initially induces a mild inflammatory reaction in the dental pulp, with this reaction gradually being replaced by healthy tissue.
Properties
It is known that these cements are bioactive in that they induce pulpal cell proliferation and cytokine release which results in hard tissue formation and the synthesis of an interface with dentine similar to hydroxyapatite composition.30 They are also extremely biocompatible with low risk of pulp or tissue reaction reported.31,32 The pH of tricalcium silicate materials is about 8.2 with the set material exhibiting a similar compressive strength, hardness and flexural modulus as dentine. Its dimensional stability is high and it has a low fluid uptake and sorption. The marginal adaptation to the cavity walls is excellent with the formation of a good seal and, in fact, its microleakage resistance is better than resin-modified glass ionomer cement. It is designed to be placed both on dentine and vital pulp tissue with plugs of the material having been observed in dentinal tubules. Biodentine™ (Septodont) has been shown to form an interfacial interaction via a dynamic mineral interaction zone with dentine. Watson et al, in 2014, examined local bioactivity between Biodentine™ (Septodont) and the subjacent dentine substrate.33 This group of workers found that the material was able to produce mineralization which extended deep into this tissue. They attributed the local ion-rich alkaline environment for the effect which seems to be more effective than that gained with the acidic-based glass ionomer cements. This offers potential for the minimally invasive treatment of carious dentine. The radiodensity of Biodentine™ (Septodont) is the same as dentine (Figure 6).
TheraCal LC (Bisco Dental Products, Schaumburg, Illinois, USA) is a tricalcium silicate cement but has been modified by the addition of resin to permit light-curing and affect other properties such as calcium ion leaching, which is lower than seen in Biodentine™ (Septodont).34
The intracoronal indications for the use of Biodentine™ (Septodont) are that it may be used as a dentine substitute (under resin composite as this restorative material may be bonded to the cement),30 a lining material in deep cavities and where a pulpal exposure is encountered either during cavity preparation35 or following trauma. Laurent et al also recommended its use for pulpotomy in primary molars.35
There is now a growing body of evidence to support use of these materials as pulpal-capping lining materials. There are a number of articles which compare the treatment outcome of mineral trioxide aggregate and setting calcium hydroxide cement as direct pulp-capping materials. Mente et al found that MTA appeared to be more effective than calcium hydroxide for maintaining long-term pulp vitality after direct pulp-capping.36 These workers also reported that teeth that were permanently restored two or more days after the pulp-capping treatment had a significantly worse prognosis in both groups and so they recommended that teeth should be definitively restored as early as possible. In 2013, Hilton et al performed a randomized clinical trial in a dental practice and found that the probability of failure at 24 months was 31.5% for the setting calcium hydroxide cement compared to 19.7% for MTA.37 Zhu et al conducted a systematic review and meta-analysis to determine the effect of mineral trioxide aggregate and setting calcium hydroxide cement for direct pulp-capping.38 It was found that MTA was superior to calcium hydroxide, resulting in a lower failure rate. Another meta-analysis was published in the same year39 and corroborated that MTA has a higher success rate and results in less pulpal inflammatory response and more predictable hard dentine bridge formation than setting calcium hydroxide cement. However, despite the initial encouraging results, it is currently too early to say whether they can be confirmed in the longer term.40 The management of a vital asymptomatic tooth in contemporary practice would therefore involve leaving affected dentine overlaying the pulp if there was any risk of exposure and using a therapeutic material such as Biodentine™ (Septodont) in an attempt to facilitate pulpal healing.
Example of a case in which Biodentine™ (Septodont) was used
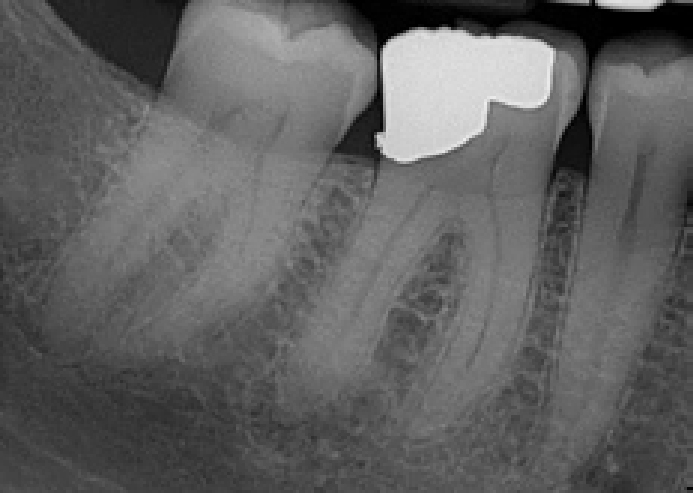
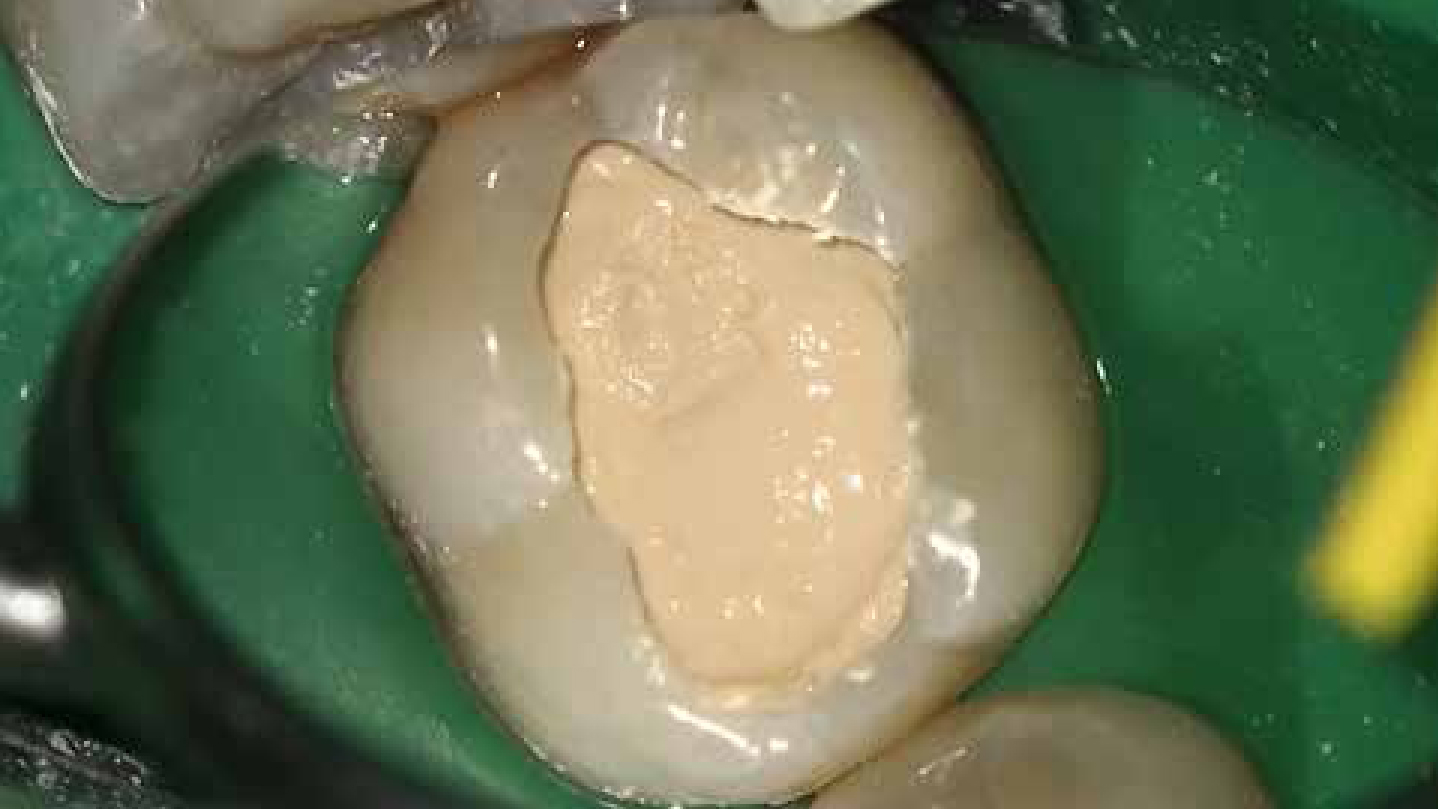
A 19-year-old female presented as a new patient. Clinically there was a shadow in LL6 consistent with a carious lesion but no cavitation of the enamel was evident. A bitewing radiograph was taken to examine the patient for any approximal lesions (Figure 7). From this radiograph, it was judged that some pulpal involvement was likely due to the potential extent of the lesion and so, after the administration of local anaesthetic, a rubber dam was placed to control moisture and, more importantly, create a controlled environment from a bacteriological perspective. The cavity was prepared with the margins cleared and the infected carious dentine removed leaving some affected caries over the pulp. Biodentine™ (Septodont) was mixed and placed in the cavity (Figure 8). The clinician has a choice of placing the material as a dressing, in which case the patient should be recalled some weeks later, for the outer part of the material to be reduced so that a definitive restorative material may be placed over it. Alternatively, the material may be placed as a lining material and covered with resin composite or dental amalgam at the same appointment. This latter approach is considered to be preferably as Barthel showed that the placement of a definitive restoration within the first two days after pulp exposure contributed significantly to an increased pulpal survival rate.41 As the patient was about to embark on some protracted medical treatment, the latter approach was chosen and the cavity was definitively restored with a resin-based composite restorative material once the Biodentine™ (Septodont) had reached full set. Clinical and radiographic follow-up has been planned to monitor the vitality of the pulp.

Conclusion
In contemporary practice, the clinician is aiming to seal the dentinal tubules opened during cavity preparation in order to prevent microleakage and, by extension, pulpal inflammation. There is now no need to place a base material in the traditional sense. There are many lining materials available on the market which are placed in a thin layer on the pulpal cavity surfaces. Indeed, a material such as a dentine-bonding agent satisfies most of the ideal properties of a lining material and therefore many clinicians would, these days, only use a product of this type, especially when it is required to be used when placing a resin composite. If it is felt that microexposure of the pulp may be present, or if a pulpal exposure is seen, then a bioactive and biocompatible lining material, such as a tricalcium silicate cement, may be used to facilitate pulpal healing. The early results of studies concerning these materials are promising.
Disclaimer
The author denies any commercial interest in any of the products or companies mentioned in this article.
